Oxygen Forms Monatomic Ions With a Charge of
What is the form. Use the absolute value of the charge on each ion as the subscript for the other ion.

Session 0 Back To The Roots Naming Compounds Flashcards Quizlet
Ionization of an oxygen atom may result in O 2- which is a monatomic ion.
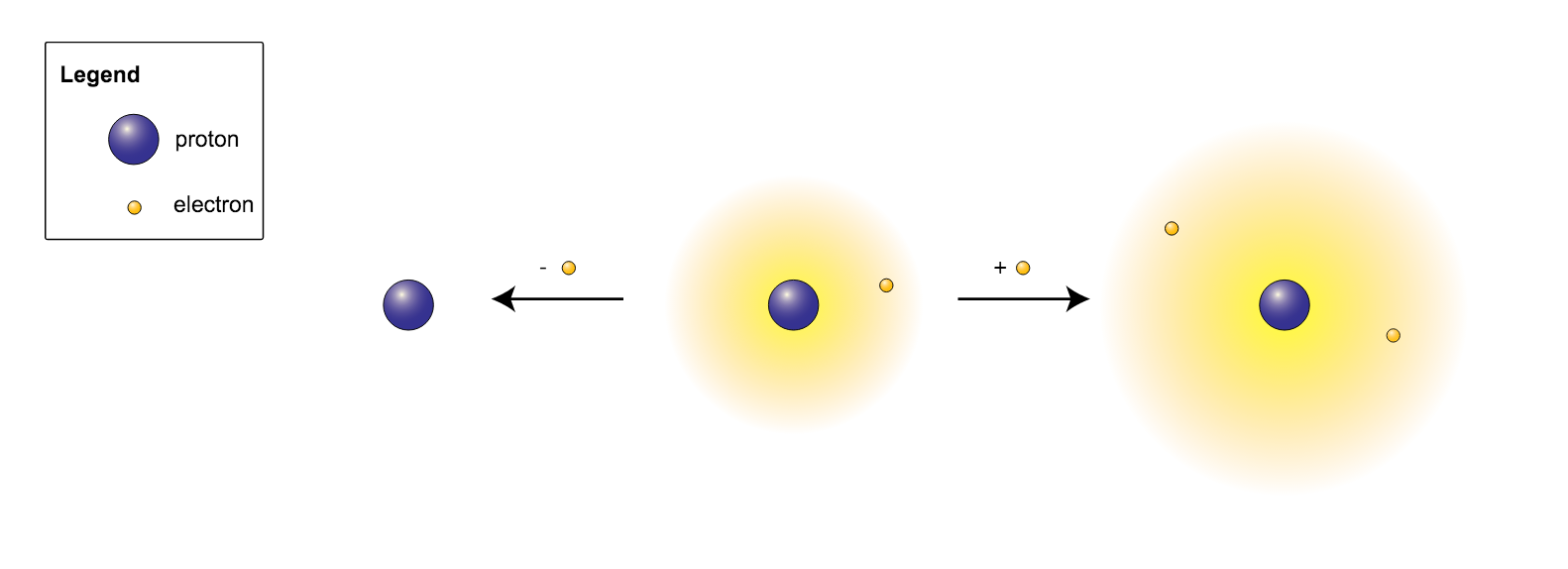
. Zn 2 zinc. It is a member of the chalcogen group in the periodic table a highly reactive nonmetal and an oxidizing agent that readily forms oxides with most elements as well as with other compoundsOxygen is Earths most abundant element and after hydrogen and helium it is the third-most abundant element in the. Hydrogen forms the only charge-1 cation that has no electrons but even cations that unlike hydrogen retain one or more electrons are still smaller than the neutral atoms or molecules from which they are derived.
H hydrogen. K potassium. Hydrogen usually forms the monatomic ion H however it sometimes acts as an anion and forms H-.
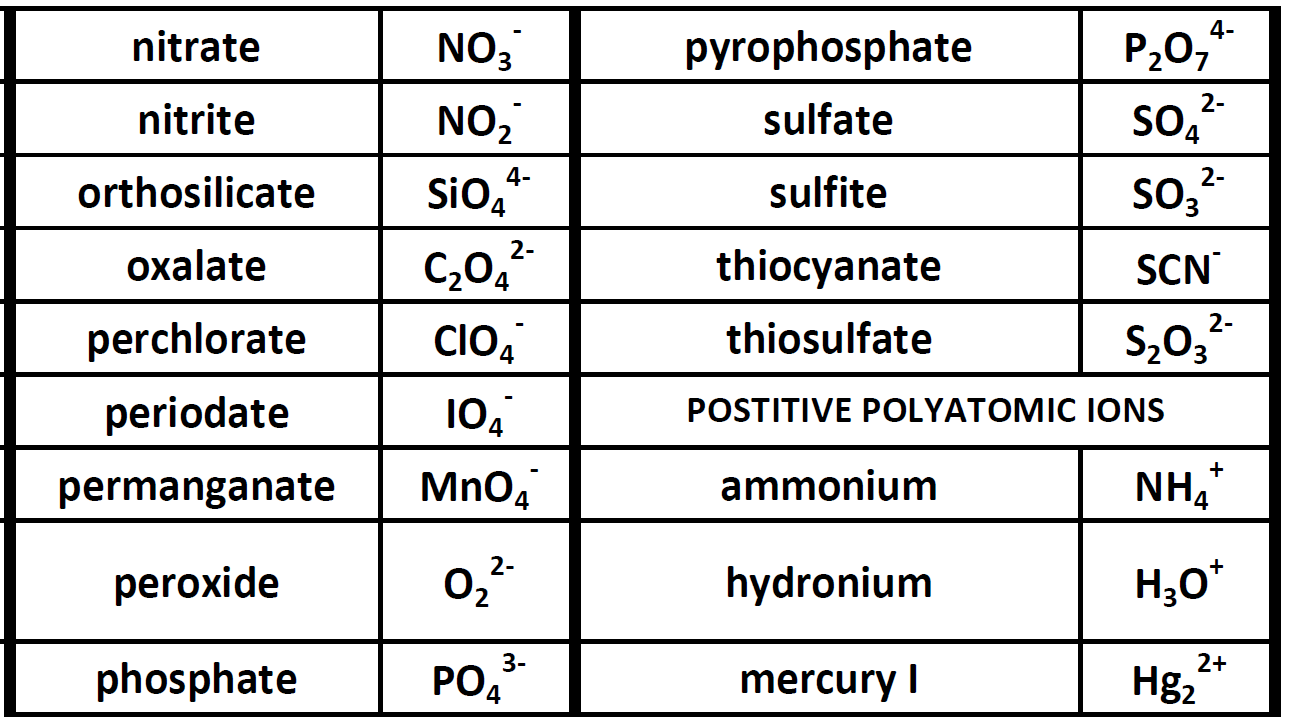
Monatomic positive ions have the name of the element from which they are formed. Since the electric charge on a proton is equal in magnitude to the charge on an electron the net electric charge on an ion is equal to the number of protons in the ion. Some of the more important polyatomic ions are listed in.
If so does it also contain oxygen. Oxygen is a colourless odourless reactive gas the chemical element of atomic number 8 and the life-supporting component of the air. They can have either a positive charge cations or a negative charge anions.
The ions that we have discussed so far are called monatomic ions that is they are ions formed from only one atom. Oxygen is the chemical element with the symbol O and atomic number 8. One of the earliest methods of distinguishing between these ions used the suffixes -ous and -ic added to the Latin name of the element to.
Each species is in its elemental form so a synthesis reaction is expected. Reduce the subscripts to the smallest whole numbers when writing the empirical formula. The monatomic ions that compose salt are sodium Na and chlorine Cl-.
The charge of an ion is always some integer value. Oxygen is a chemical element with atomic number 8 which means there are 8 protons and 8 electrons in the atomic structure. Sodium ions have a charge of 1 chlorine ions a charge of -1.
It is a member of the chalcogen group on the. Because the actual formula units are neutral. Both of these ions are monatomic ions.
It is more common for polyatomic ions to be negatively charged than to be positively. A monatomic ion with a charge of 2 has an electronic configuration of 1s22s22p63s23p64s23d104p6. Atoms or groups of atoms that possess a net electrical charge are called ions.
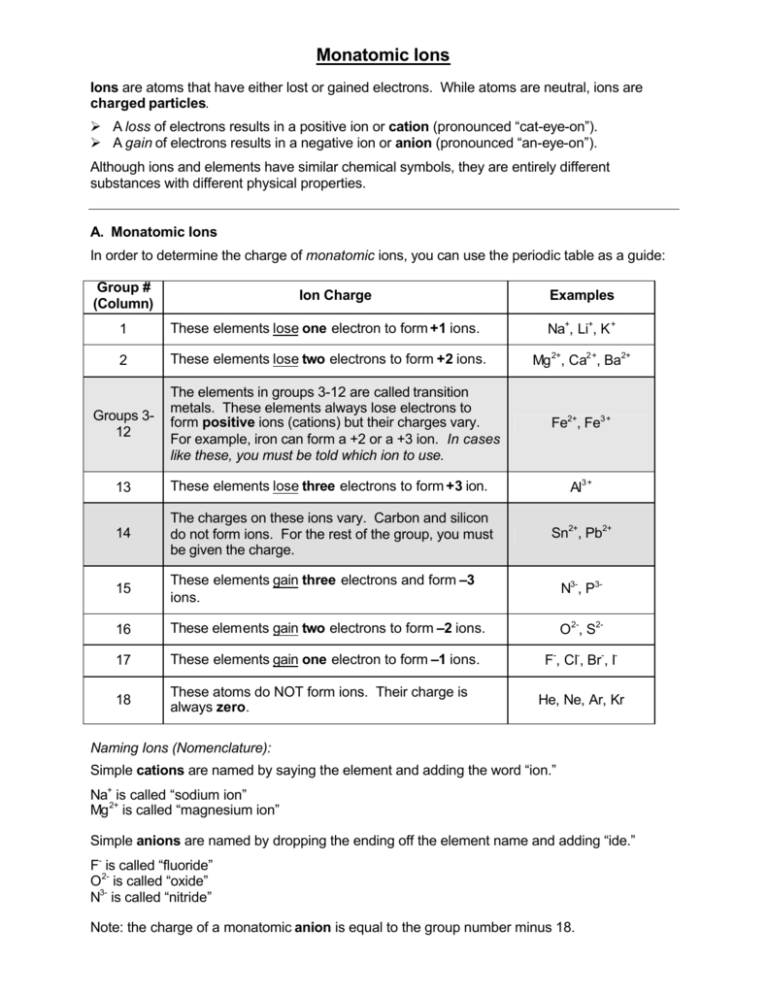
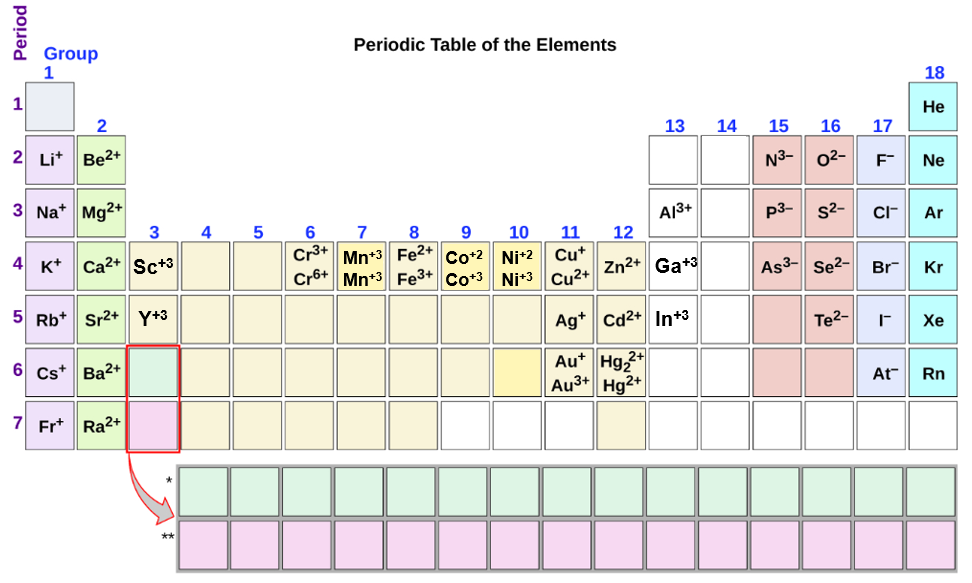
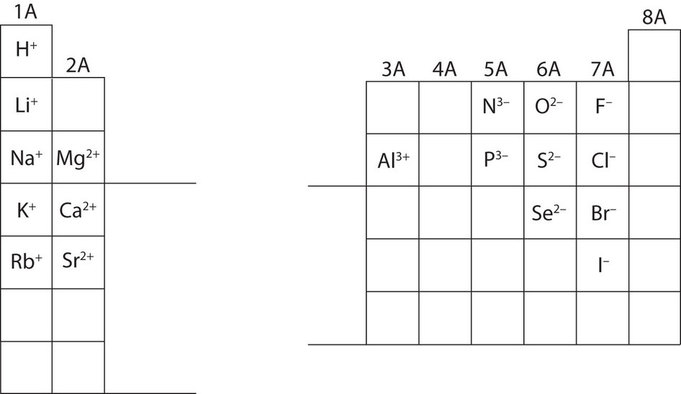
Are the ions monatomic or polyatomic. If the compound is ionic does the metal form ions of only one type fixed charge or more than one type variable charge. The charges on monatomic ions of most main group elements can be predicted from the location of the element in the periodic table.
Ions can consist of one atom monatomic ions or several polyatomic ions. Sr 2 strontium. The charge of the negative oxygen end in water is about -23e about two thirds the charge of a single electron.
Another decomposition that will generate CO2 and a metal oxide note that Al forms a 3 ion Al3 and monatomic oxygen will have a 2 charge O2-. Note that when mercury carries a 1 charge it forms an uncommon polyatomic ionic state. KCl dissociates in water into K and Cl-ions.
From the answers we derive we place the compound in an appropriate category and then name it accordingly. Some metals form positive ions in more than one oxidation state. We also find many polyatomic ions.
These ions which act as discrete units are electrically charged molecules a group of bonded atoms with an overall charge. Synthesis 4Al 3O2 2Al2O3. The chemical symbol for Oxygen is O.
All of these are monatomic ions even though they have a charge that is more than 1 because they are composed of a single atom. Bearing a charge and interacting with other ions and compounds just like the monatomic ions discussed above. What is the chemical symbol of the noble gas this ion is isoelectronic with.
Because these ions are made of multiple atoms they are called polyatomic ions. Since the most stable ionic form of aluminum is Al3 and oxygen is O2- Al2O3 will. The negative ion anion may be a monatomic non-metal ion such as Cl- or a polyatomic ion such as SO4 2-or NO 3- When writing formulas for ionic compounds the net charge of the formula unit must be zero.
Ca 2 calcium. Oppositely charged ions will come together in a ratio that makes neutral units of the. Polar molecules have partially charge dipoles and their charge value is not an integer.
Na sodium. If the compound is molecular does it contain hydrogen. Carbon As An Ion Monatomic Ions.
B Because HPO 4 2 has a charge of 2 and Na has a charge of 1 the empirical formula requires two Na ions to balance the charge of the polyatomic ion.
Ch104 Chapter 3 Ions And Ionic Compounds Chemistry

Monatomic Ion Definition And Examples

Monatomic Ions Definition Naming Convention Video Lesson Transcript Study Com

Molecular And Ionic Compounds Chemistry For Majors

Monatomic Ions Definition Naming Convention Video Lesson Transcript Study Com

Chapter 7 Ionic Compounds And Metals
2 6 Ionic Compounds And Formulas Chemistry Libretexts

Monatomic Ions Of The Main Group Elements And Their Ionic Charges

Select A Section Introduction Atoms Molecules And Ions Laws And Theories A Brief Historical Introduction 2 1 Laws Of Chemical Combination 2 2 John Dalton And The Atomic Theory Of Matter 2 3 The Divisible Atom 2 4 Atomic Masses 2 5 The Periodic
Naming Monatomic Ions And Ionic Compounds Article Khan Academy
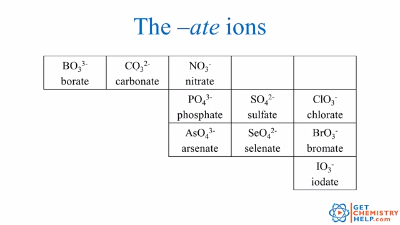
Chemistry Lesson Polyatomic Ions Get Chemistry Help
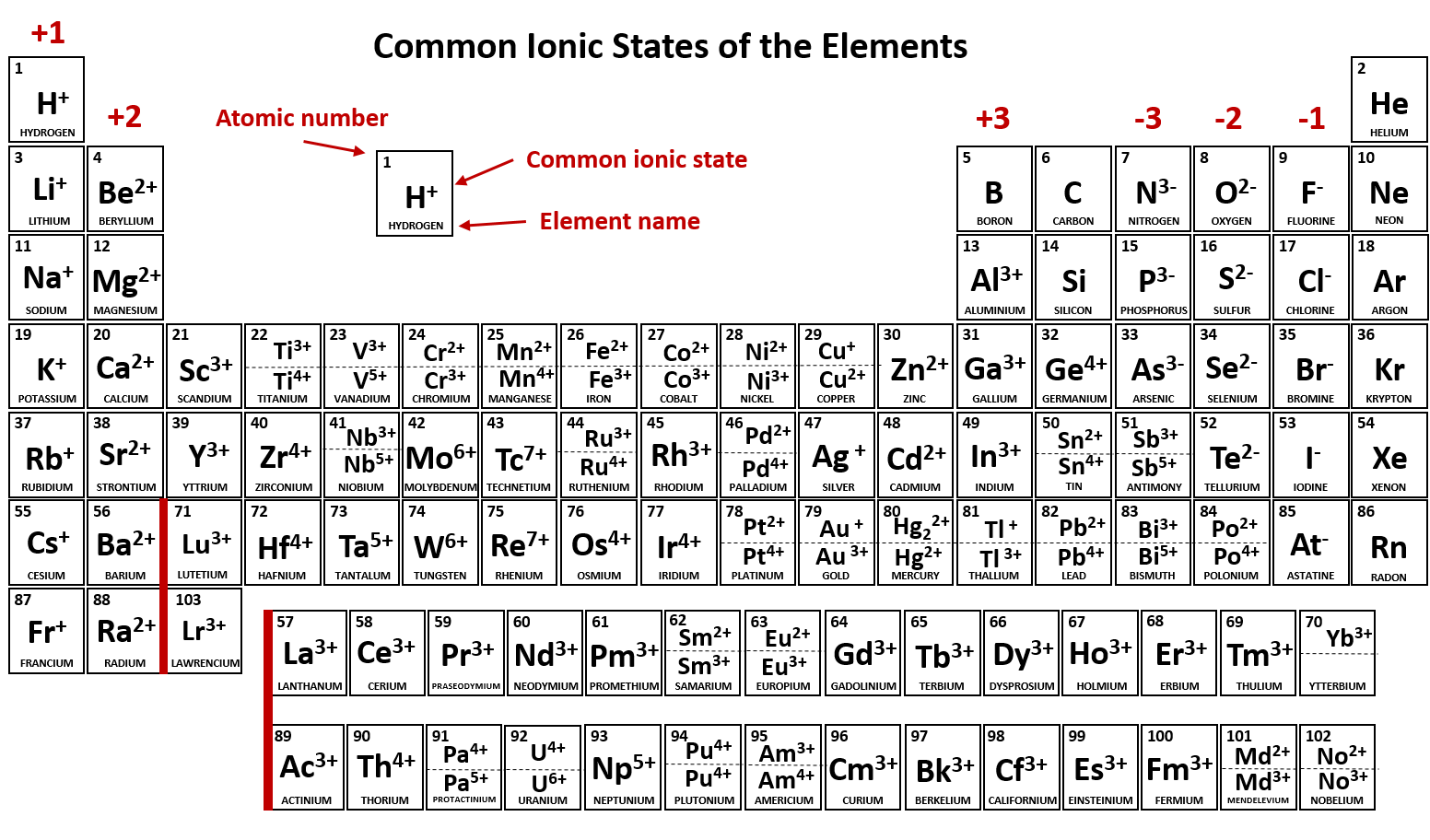
Ch104 Chapter 3 Ions And Ionic Compounds Chemistry
Why Is Oxygen In Chemical Equations Written As O2 When The Element Itself Is Just O Consider Alkane O2 Co2 H2o Energy Are Other Elements Written This Way In Chemical Equations Quora
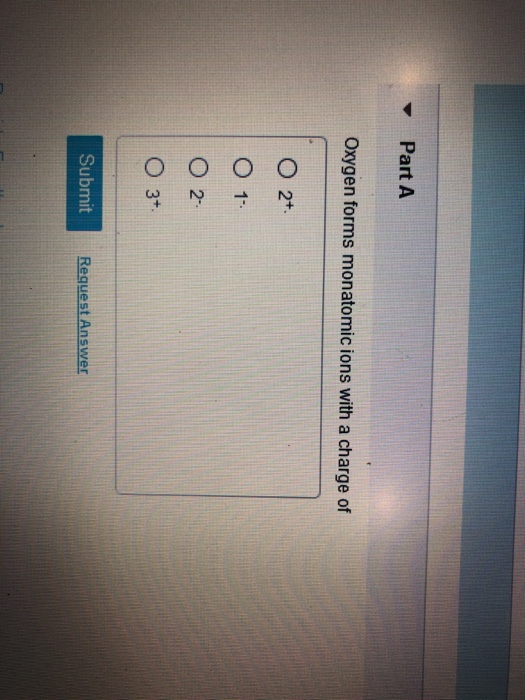
Solved Part A Oxygen Forms Monatomic Ions With A Charge Of O Chegg Com
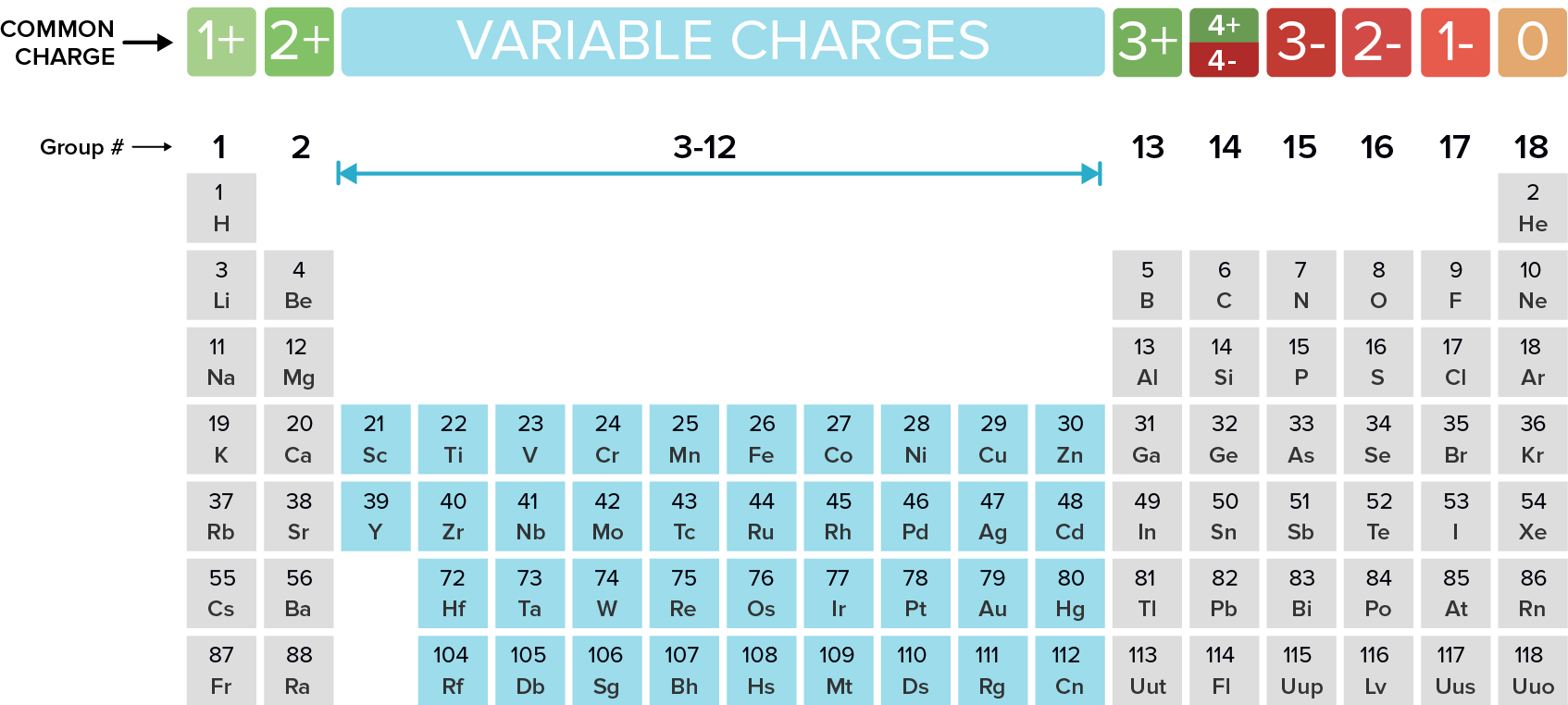
Naming Monatomic Ions And Ionic Compounds Article Khan Academy

Select A Section Introduction Atoms Molecules And Ions Laws And Theories A Brief Historical Introduction 2 1 Laws Of Chemical Combination 2 2 John Dalton And The Atomic Theory Of Matter 2 3 The Divisible Atom 2 4 Atomic Masses 2 5 The Periodic

Comments
Post a Comment